The EACR’s ‘Highlights in Cancer Research’ is a regular summary of the most interesting and impactful recent papers in cancer research, curated by the Board of the European Association for Cancer Research (EACR).
The list below appears in no particular order, and the summary information has been provided by the authors unless otherwise indicated.
Use the dropdown menu or ‘Previous’ and ‘Next’ buttons to navigate the list.
Chia, S. B., Johnson B. J. et al. Nature. 645: 496–506. (2025).
doi: 10.1038/s41586-025-09332-0.
Summary of the findings
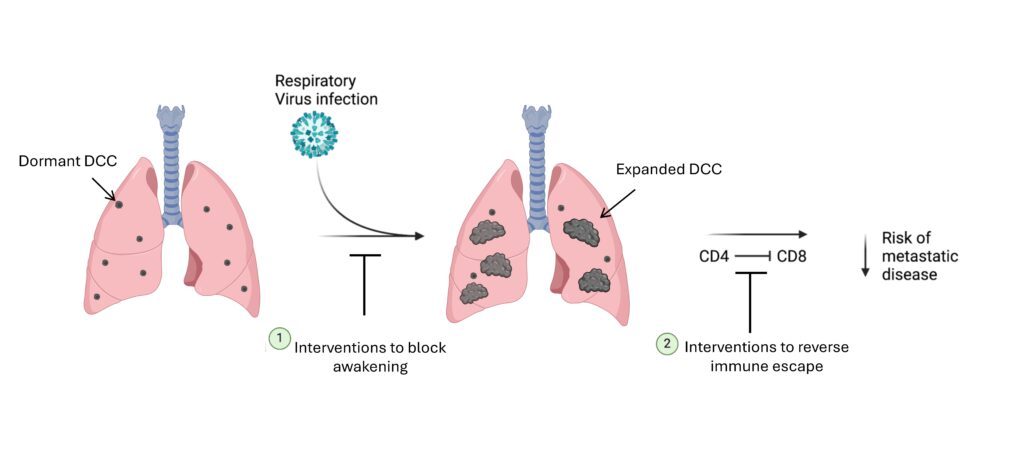
Cancer cells which disseminate from primary tumors (disseminated cancer cells, DCC) can remain dormant and persist in target organs up to decades before awakening to form overt metastatic disease. The DCC microenvironment determines dormancy or awakening, and acute respiratory viral infections (RVI) like SARS-CoV-2 and influenza virus induce potent inflammation in the lungs. Our recent studies demonstrated in mice that influenza A virus (IAV) and SARS-CoV-2 infections lead to loss of the pro-dormancy phenotype in breast DCC in the lung, causing DCC proliferation within days of infection, and a massive expansion of carcinoma cells into metastatic lesions over a few weeks. These phenotypic transitions (altered expression of epithelial/mesenchymal markers) and expansions are interleukin-6 (IL-6) dependent.
Later in the infection, DCC co-localize with CD4+ T cells in iBALT (induced bronchioalveolar lymphoid tissue), with few CD8+ T cells. While depleting CD4+ T cells did not affect the initial DCC expansion post-IAV infection, CD4+ T cell depletion led to an almost 100X reduction in DCC burden by 28 dpi. Importantly, depletion of both CD4+ and CD8+ T cells reverses the effect of depleting only CD4+ cells, indicating that CD4+ T cells were interfering with anti-tumor responses mediated by CD8+ T cells (further demonstrated by cytotoxic T-lymphocyte killing assays ex vivo). Using single cell (sc) RNAseq, we further showed that DCC can reprogram CD4+ and CD8+ T cells, with gene expression profiles consistent with immune suppression. Thus, CD4+ T-cells sustain pulmonary metastatic burden post-RVI by interfering with CD8+ T-cell cytotoxicity.
These experimental findings align with human observational data. Analyses of cancer survivors from the UK Biobank (all cancer) and Flatiron Health (breast cancer) databases revealed an increase in risk of cancer-related mortality and lung metastasis, respectively, in cancer survivors who suffered from SARS-CoV-2 infection when compared to those uninfected. These discoveries highlight the significant impact of RVI on metastatic cancer resurgence, offering novel insights into links between RVI and cancer risks.
.
.Future impact
Key goals moving forward will be to extend these discoveries to reduce the risk of metastatic progression in cancer survivors who experience RVI. The most obvious, and least risky, approach will be to prevent the RVI in the first place, such as through avoiding exposures (e.g. masking) or through vaccination. For vaccination, we will compare different vaccination strategies for their ability to effectively prevent dormant DCC awakening and metastatic disease progression. Additional studies will seek to develop interventions that could be applied during or after infection, with the goal of blocking the pathways that mediate awakening (i.e. the IL-6 pathway) or that mediate immune escape. In all, our goal will be to capitalize on mechanistic understanding of RVI induced metastatic progression to limit its impact on cancer survivors.