The EACR’s ‘Highlights in Cancer Research’ is a regular summary of the most interesting and impactful recent papers in cancer research, curated by the Board of the European Association for Cancer Research (EACR).
The list below appears in no particular order, and the summary information has been provided by the authors unless otherwise indicated.
Use the dropdown menu or ‘Previous’ and ‘Next’ buttons to navigate the list.
Heide, J. et al. Nature. 645:1051-1059. (2025).
doi: 645(8082):1051-1059.
Summary of the findings
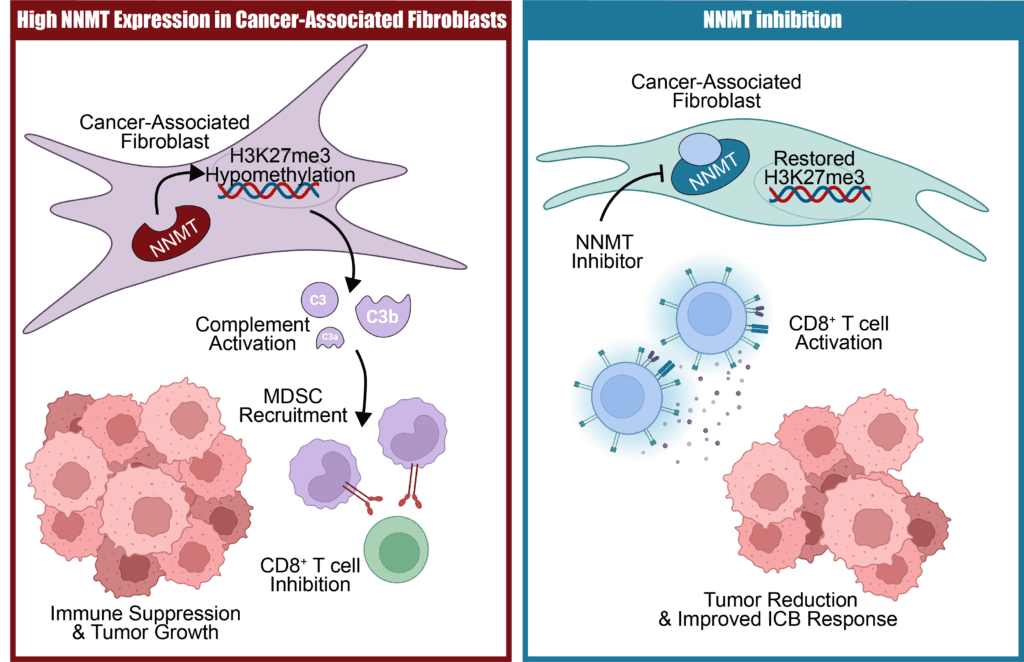
Nicotinamide N-methyltransferase (NNMT) is a metabolic and epigenetic regulator that reprograms normal fibroblasts into cancer-associated fibroblasts (CAFs) across various types of cancer (Eckert et al., 2019). Mechanistically, high NNMT expression in CAFs depletes the universal methyl donor S-adenosylmethionine (SAM) to produce 1-methylnicotinamide, creating a metabolic sink. This decrease in SAM availability results in a global loss of H3K27me3, an epigenetic change that encourages a pro-tumorigenic CAF phenotype.
NNMT-activated CAFs create an immunosuppressive tumor microenvironment by secreting complement factors. Activation of the complement cascade activation recruits Ly6Chigh monocytes to the tumor site and promotes their differentiation into PD-L1+ monocytic myeloid-derived suppressor cells (M-MDSCs). These MDSCs directly inhibit the proliferation and effector functions of CD8+ T cells, thereby shielding the tumor from antitumor immunity. Nnmt knockout in the non-immune stroma of immunocompetent mice significantly reduces tumor growth and restores CD8+ T cell activity in syngeneic models of ovarian, breast, and colon cancer.
To translate these findings into a therapeutic strategy, we developed a potent, specific, and orally bioavailable NNMT inhibitor (NNMTi). X-ray crystallography revealed that this inhibitor effectively occupies both the cofactor- and substrate-binding pockets of the enzyme. In vivo, NNMTi treatment reverses the epigenetic reprogramming of CAFs, reduces C3 secretion, and decreases the recruitment of immunosuppressive M-MDSCs, while increasing CD8+ T cell activation.
Consequently, NNMTi treatment decreased tumor burden and metastasis in multiple mouse models. We further show that NNMTi treatment sensitized tumors to immune checkpoint blockade, such as anti-PD-1 and anti-CD47, by restoring CD8+ T cell activation. These findings establish NNMT as a key epigenetic regulator of CAF activation and a promising therapeutic target to reduce stroma-driven immunosuppression and enhance the effectiveness of immunotherapies.